ABOUT
Caywon Pharmaceutical Group, Ltd. was founded by a group of seasoned pharma/healthcare IP attorneys, making it a rare, highly-specialized team operating in the field of pharmaceutical product development. Caywon�s distributed partnering approach to pharmaceutical product development provides a unique global platform for business entities to benefit from advances in modern science and technology through innovative transactional and commercialization programs. Caywon has subsidiaries in the United States., China, and Hong Kong.
Our Mission
At Caywon, we believe that access to affordable and quality healthcare should be a basic human right regardless of national borders. In many countries, access to the latest medicine is a privilege only available to those who can pay the price. In the 21st century, the biggest challenge facing healthcare is not the lack of medical innovations but the inefficient model of product developments and healthcare delivery. Our mission is to seek out business models that can accelerate the translation of new medical innovations to the marketplace and to develop new technologies that will help deliver quality healthcare to all at affordable prices. Simply put, we seek to democratize healthcare and make it a basic right to all.
Digital First
76% of patients think that pharmaceutical companies have a responsibility to provide information and services that help patients to manage their health. Customer engagement has been the central theme in the digital transformation of many industries. However, the pharmaceutical industry as a whole has been far behind the curve in its digital transformation. Since our founding, Caywon has been at the forefront of business model innovation and built our operations from the ground up to be a �digital first� organization. From being one of the first drug companies to take advantage of China�s dual-filing regulatory framework to being the first company to adopt the revolutionary Crowd Machine technology to create a decentralized drug development and commercialization platform, we are leading the industry in embracing the decentralized digital future.

RESEARCH & DEVELOPMENT
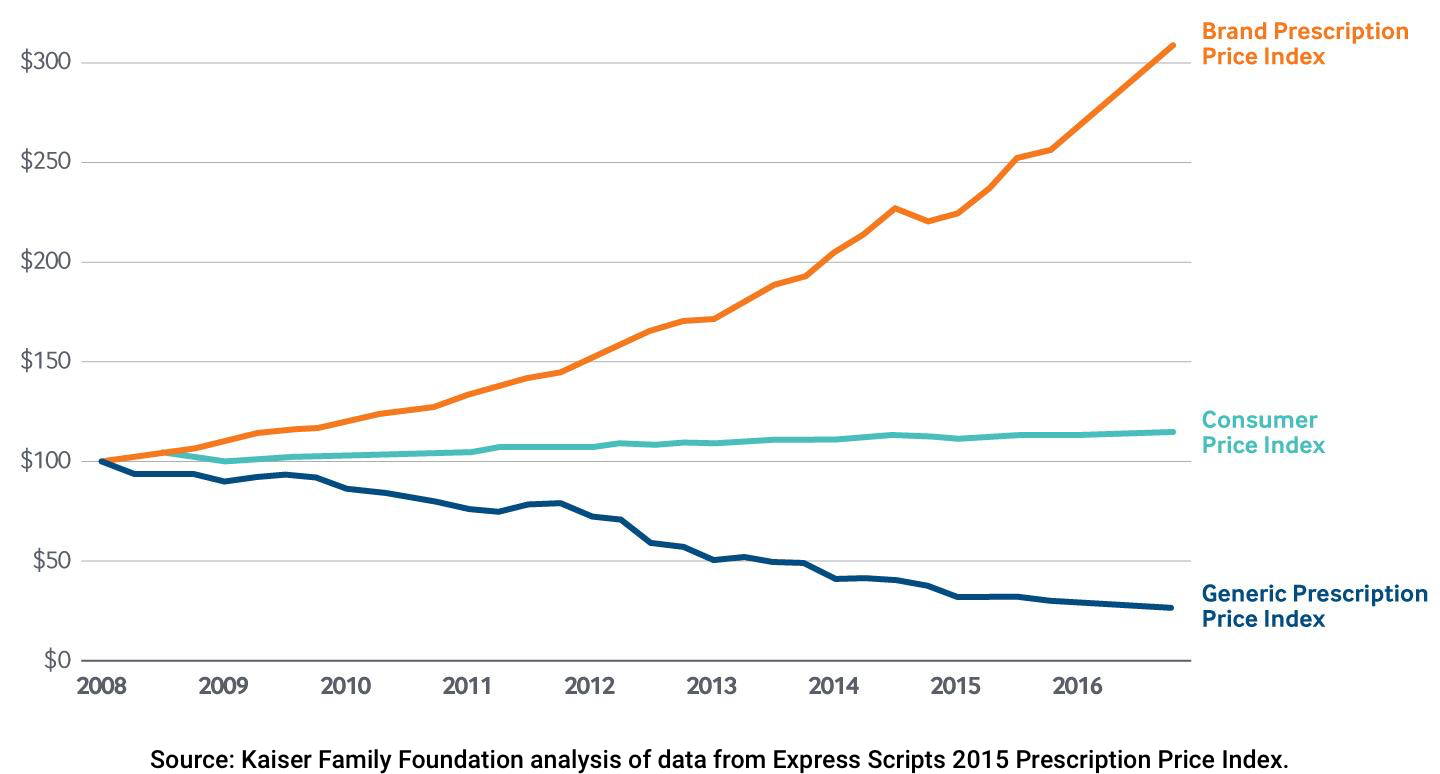
FIRST GENERICS & SUPER GENERICS
Price for brand-name prescription drugs doubled between 2008 and 2016, while about one third of the rise was a result of either prices increases for drugs or a shift in prescribing toward higher-price products. The rising drug prices has created barriers to healthcare access that affect patients, providers and payers. According to a recent study by the Commonwealth Fund (June 2017), a big contributor to the rising cost of drugs has been the high launch costs.
Caywon�s team of IP attorneys and product definition experts worked diligently to develop a new model that has a more efficient cost structure for the development of generic pharmaceutical products. This new model will allow us to introduce quality generic drugs with competitive prices to the marketplace sooner. At Caywon, we also focus our attention on improving existing drugs and finding new uses for drugs that we already know is safe for human consumption.
Our pipeline of drugs include candidates in cardiovascular, endocrinology, gastroenterology, immunology, neurology, rheumatology, digestive and sleep disorder.
INNOVATIVE DRUG
With the popularity of mobile devices, myopia has reached an epidemic proportion. Throughout the world, myopia prevalence ranges from 8% to 62%. It is projected that the prevalence of myopia will only increase if left unchecked. Currently, there are no approved medications for treating myopia. The only options are glasses, contacts, or surgery, and the only effective medication known to the medical community, atropine, has significant side-effects that renders it impractical as a medication. Through Caywon�s distributed partnering network, we have identified a new opportunity for treating myopia through medicinal means. To bring this opportunity to the market, Caywon is actively researching and developing crowd-sourcing models to help generate the data needed for clinical development. This will be an exciting new approach to medical product development that fully utilizes distributed partnering in a �shared economy� model in which the cost of drug development is significantly lowered and the benefit is shared across all stakeholders.


DIGITAL FIRST & DISTRIBUTED PARTNERSHIP
Traditional models of drug development in pharma and biotech have both reached a point of diminished return. As the cost of developing new drugs increases, many promising early stage drugs are not pursued because they represent too much risk to investors and traditional drug developers. To combat the out-of-control cost, Cawyon is among the first generation of forward thinking companies that is adopting a distributed partnering model to pharmaceutical development. By utilizing modern social network technologies and distributed computing to divide and distribute the tasks needed to be done to a network of service providers, the risks of drug development can be externalized to achieve a drastically reduced cost structure for developing new pharmaceutical products. This digitally-enabled �sharing economy� principle lies at the heart of Caywon�s digital first distributed partnership model.
BLOCKCHAIN SOLUTIONS IN HEALTHCARE
Touted as the third-generation internet, blockchain has emerged as a truly disruptive digital technology that is poised to revolutionize human commerce as we know it. At Caywon, we are actively researching and developing blockchain applications in healthcare. Our recent partnership with Crowd Machine will further accelerate our development of practical solutions to address some of the most challenging problems facing healthcare today, ranging from supply chain integrity to virtual value chain management and digital security of healthcare information and facilities.

PRESS
-
Alt Coin Latest News
Blockchain innovation to outfit researchers as well as medicine designers (Includes meeting as well as first-hand account)
In enhancement to having the ability to create blockchain applications with zero-code, researchers could create simulations to work on the international network of computer systems.
[Read more...]
-
Coinmis
Caywon Pharmaceuticals Group uses blockchain technology to develop group machines to ensure data security and integrity
Caywon Pharmaceuticals uses blockchain technology to equip scientists with the IT tools necessary to digitize their needs with the Crowd Machine work process.
[Read more...]
-
Irish Manufacturing
Blockchain technology fast tracking drug R&D and manufacturing
A lot has been written about the benefits of blockchain technology in the pharmaceutical industry, especially in regards to the supply chain - tracking and stopping counterfeit drugs.
[Read more...]
-
Pharma Voice
Blockchain Technology
Blockchain can be a challenging concept to get your head around, and it�s difficult to embrace something you can�t understand.
[Read more...]
-
Process Worldwide
Caywon Reveals Pharma 4.0 Digital Transformation Initiative
Caywon Pharmaceutical Group will become the first international pharmaceutical company to adopt Crowd Machine as the key enabling technology to power its Pharma 4.0 digital transformation initiative.
[Read more...]


